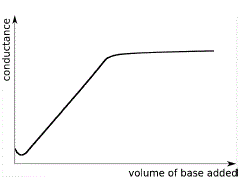
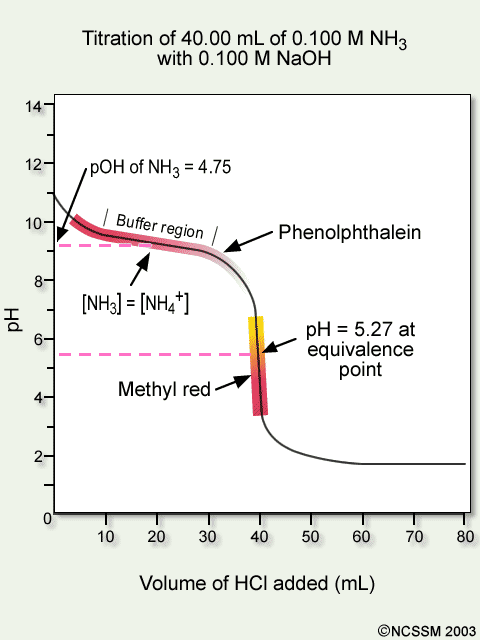
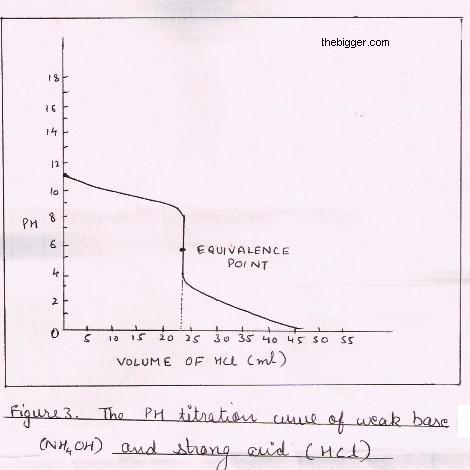
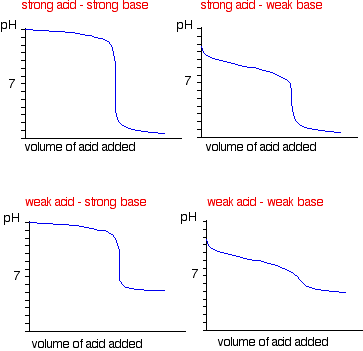
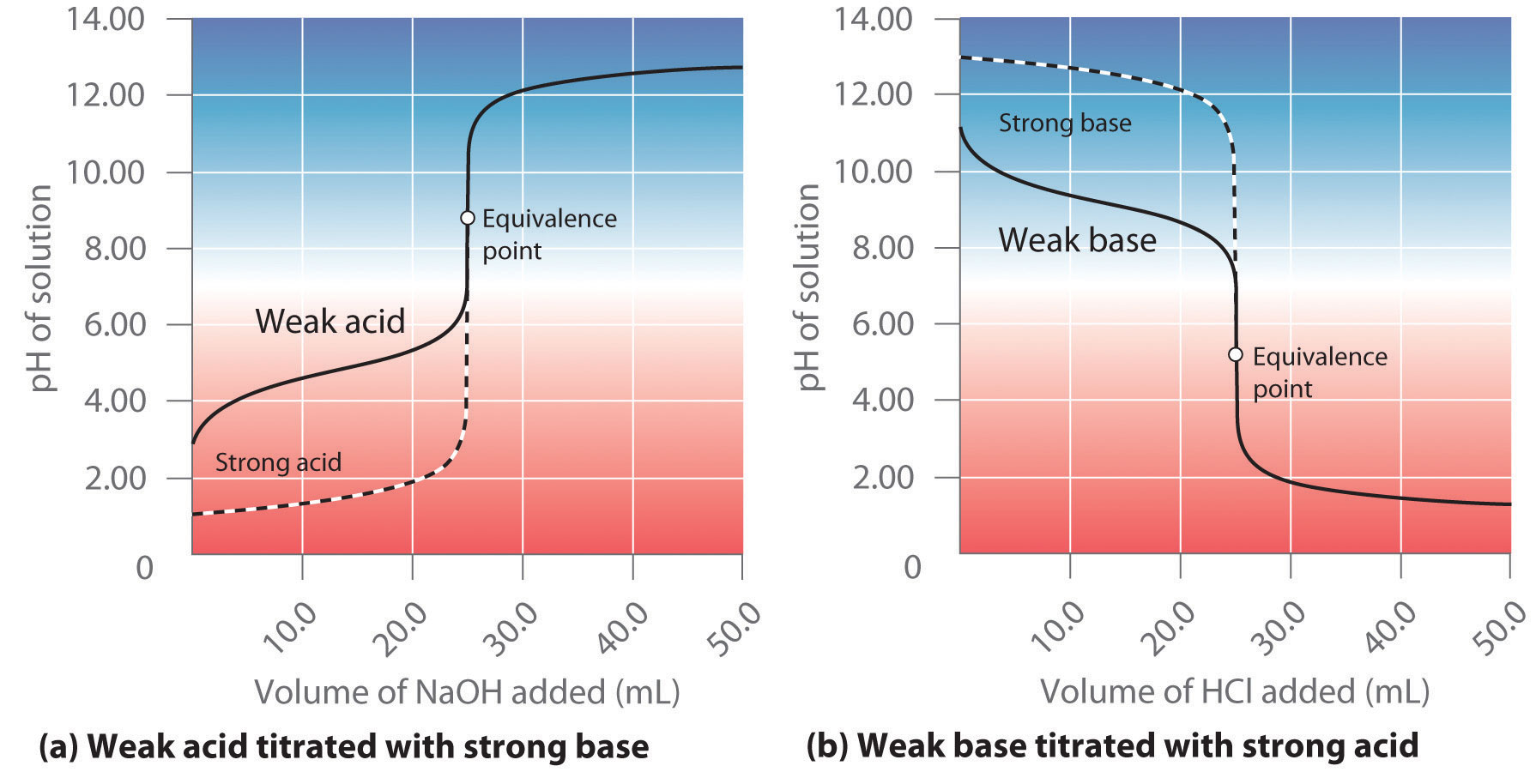
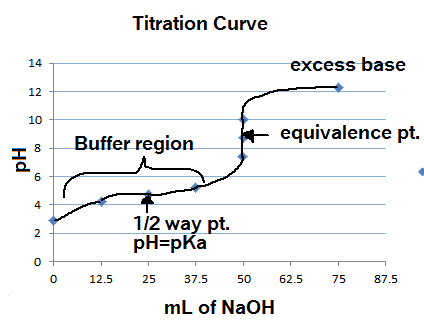
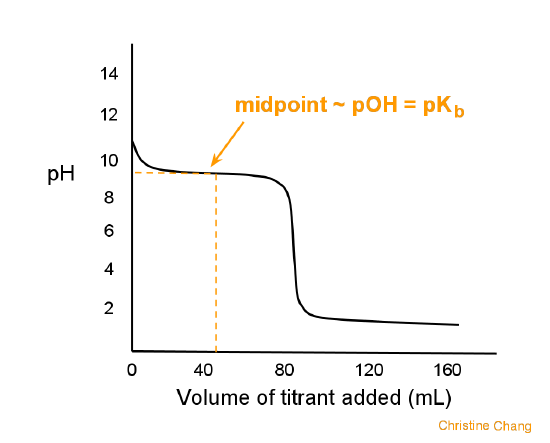
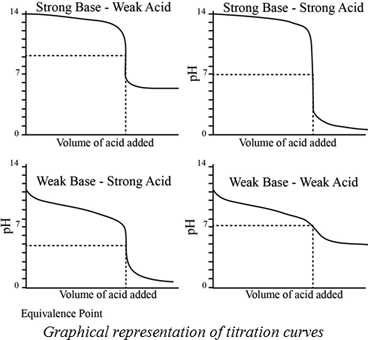
Conductometric titration of strong acid and weak base (strong acid vs weak base) - video Dailymotion
NaHCO 3 is titrated to CO 3 2- with NaOH ; an indicator used has to be suggested. Concept introduction: A titration is one of the most useful ways of determining accurately