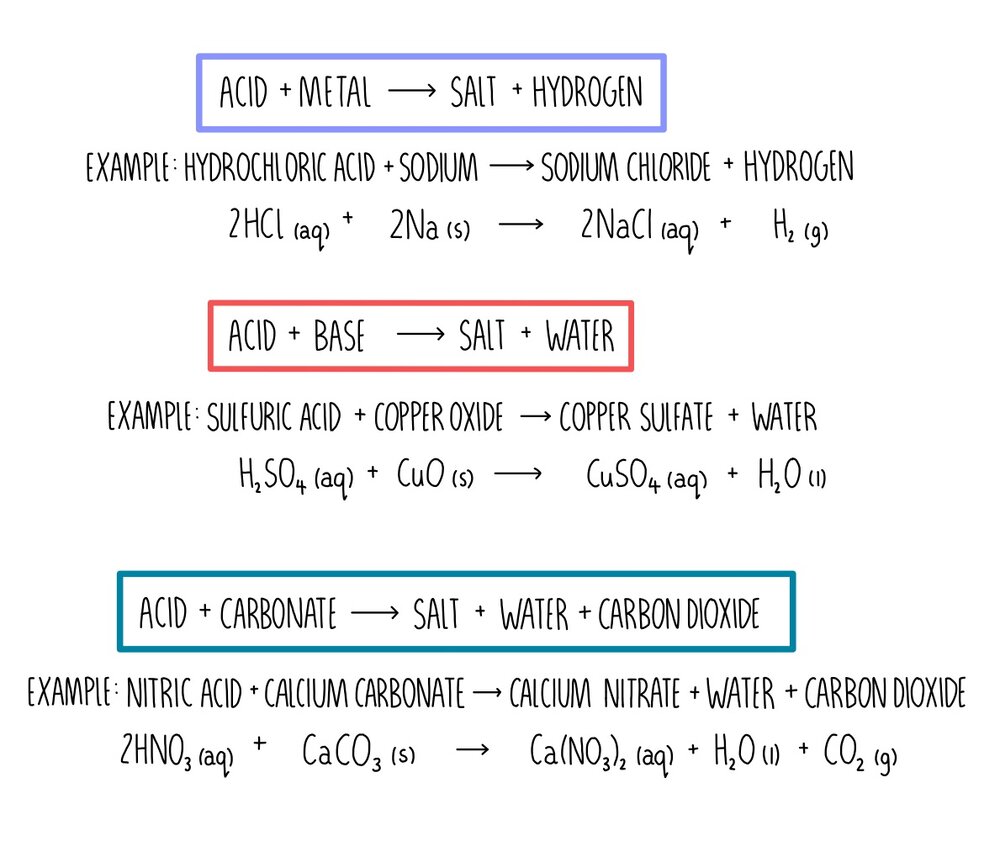
Reactions. DON'T COPY pink WRITING. Acid + base This is a common reaction and needs to be remembered for exams. ACID + BASE -> SALT + WATER E.g. hydrochloric. - ppt download

Write a mechanism (using curved-arrow notation) for the deprotonation of tannins in base. Use Ar-OH as a generic form of a tannin and use sodium carbonate (Na2CO3) as the base. Balance the