The Nernst equation giving dependence of electrode reduction potential on concentration is - YouTube

The standard reduction potential for `Cu^(2+)|Cu` is `+0.34V`. Calculate the reduction potential... - YouTube
2 The standard half reduction potential of Ag+|Ag is 0.79V is 25^° C. Given the experimental value Ksp=1.5 10* 10 for AgCl, calculate the standard half cell reduction potential for the Ag|AgCl
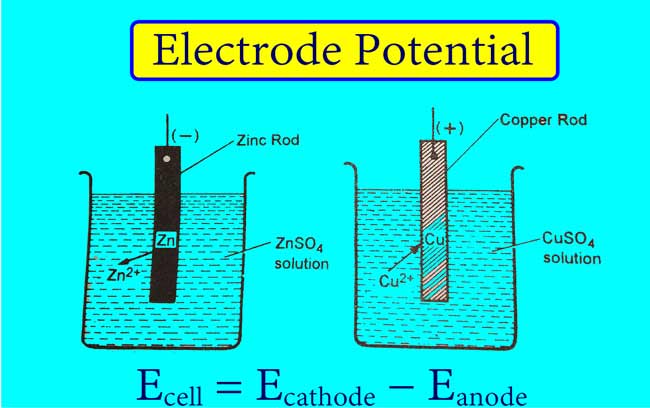
Electrode Potentials and Electrochemical Cells - Electrode Potentials (A-Level Chemistry) - Study Mind

Give Nernst equation:Calculate the electrode potential of the following single electrode. Cu(aq)^++ (C = 0.01M)/ Cu; (E^∘ = + 0.337 V)

Half-cell potentials Electrochemical Series using E cell predicting reaction feasibility A level GCE AS A2 chemistry revision notes KS5

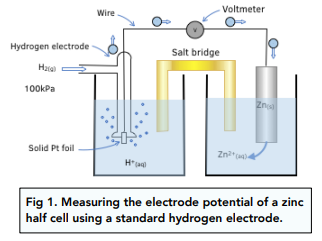
How to find the electrode potentials of hydrogen electrodes at 25 °C and 100 kPa in solutions of pH 0 and pH 14 - Quora

Given standard electrode potentials: Fe^3 + + 3e^-→ Fe;E^0 = - 0.036V Fe^2 + + 2e^-→ Fe;E^0 = - 0.440V The standard electrode potential E^o for Fe^3 + + e^ - → Fe^2 + is:

Calculate the standard electrode potential of the Ni^(2+)//Ni electrode , if the cell potential ... - YouTube