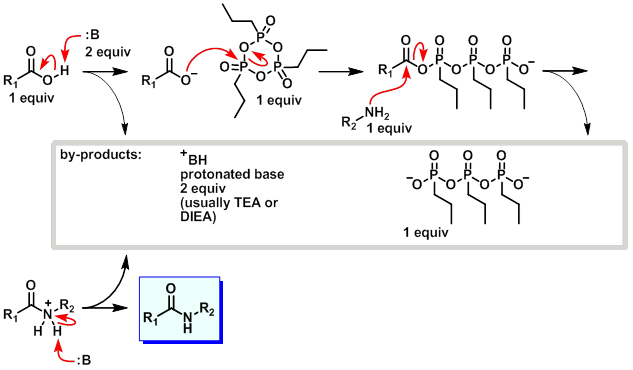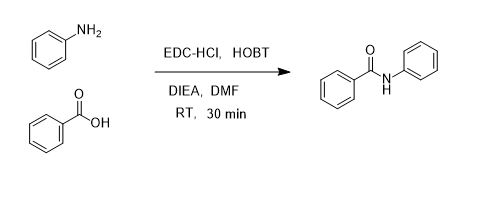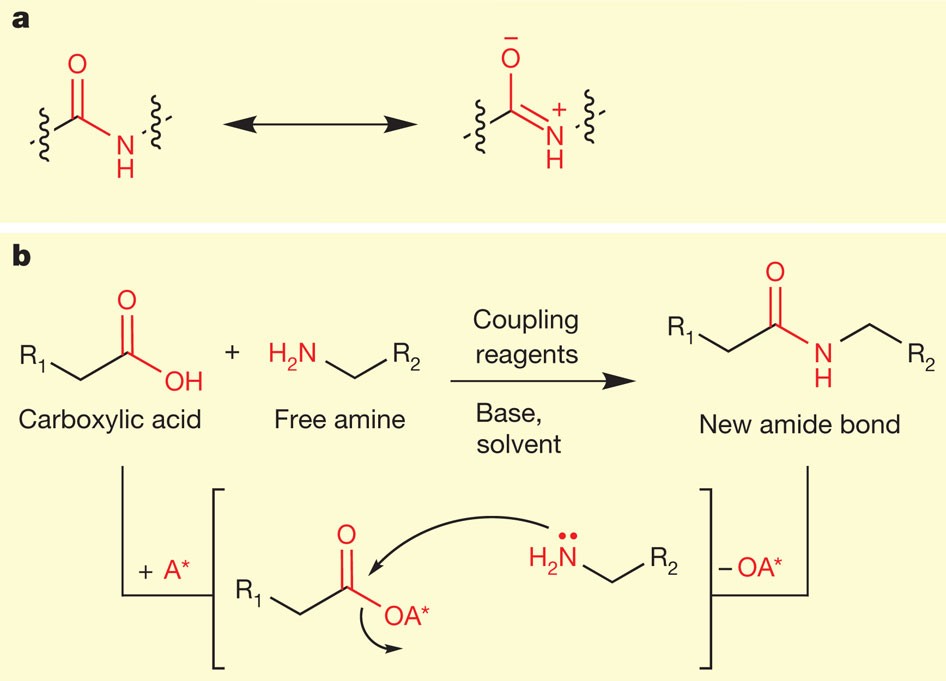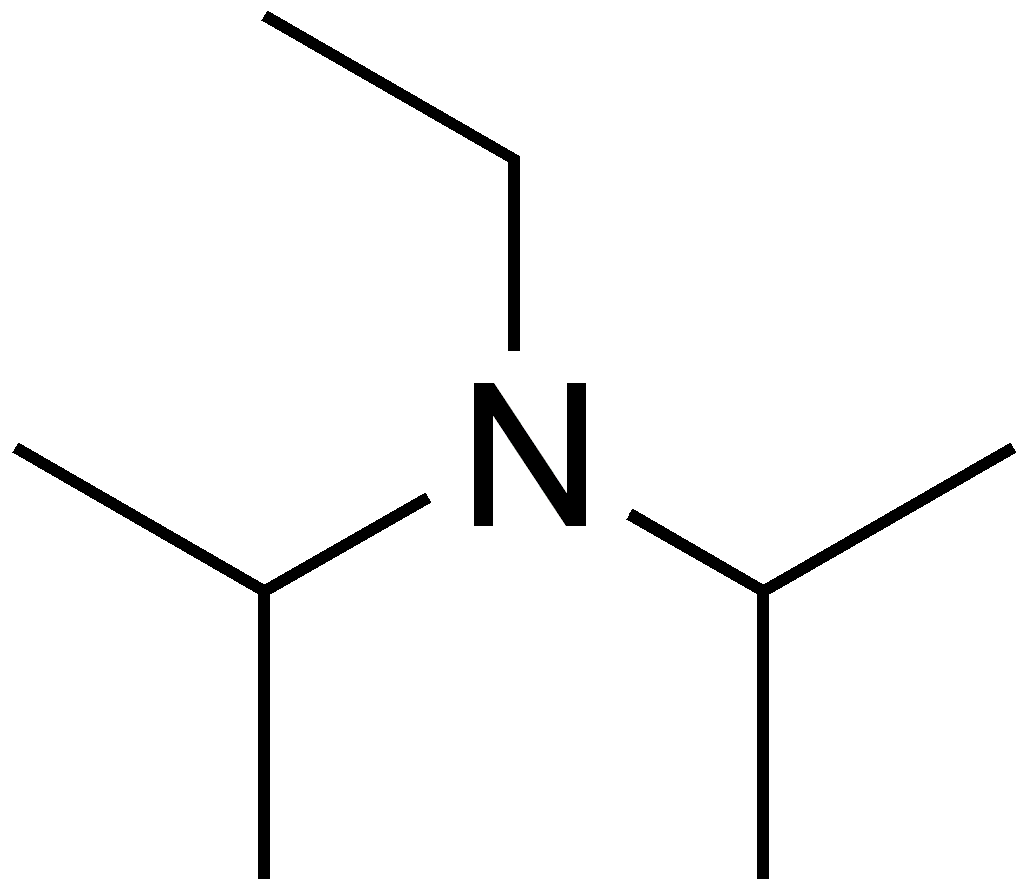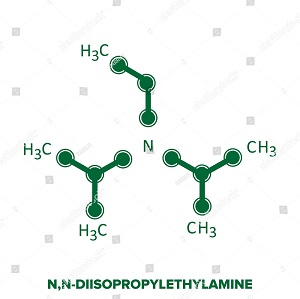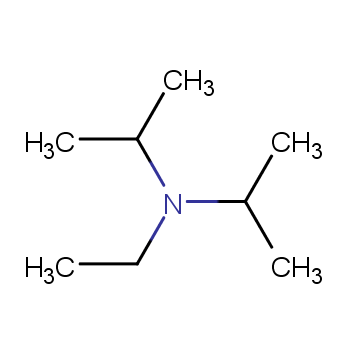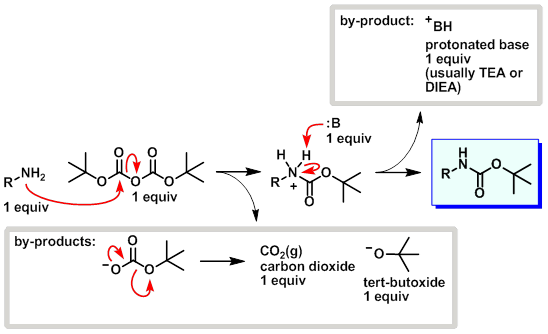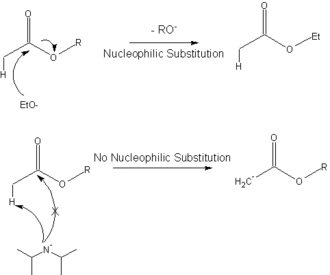
7087-68-5 | N,N-Diisopropylethylamine | 1,1'-Dimethyltriethylamine; Bis(1-methylethyl)ethylamine; DIEA; DIPEA; Diisopropylethylamine; Ethyl-N,N-diisopropylamine; Ethyldiisopropylamine; Huenig's base; Hunig's base; Hunig's reagent; N,N-Bis(1-methylethyl ...
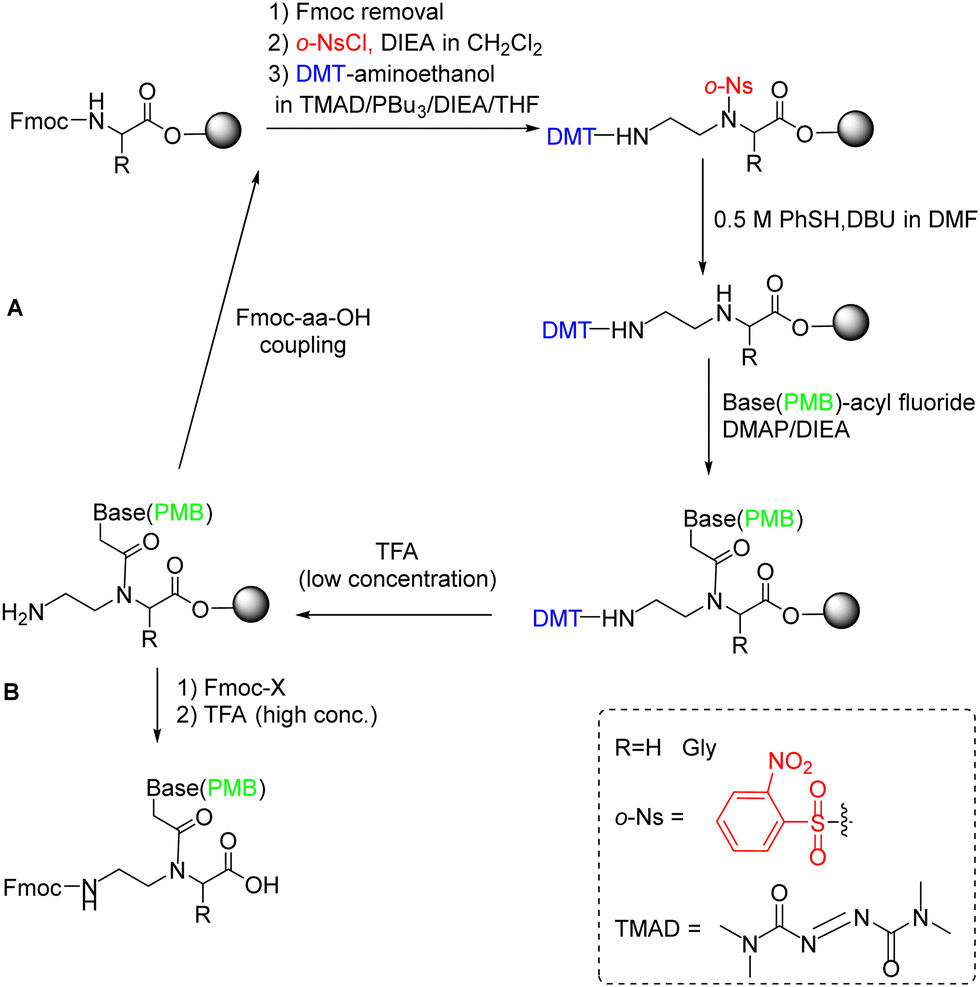
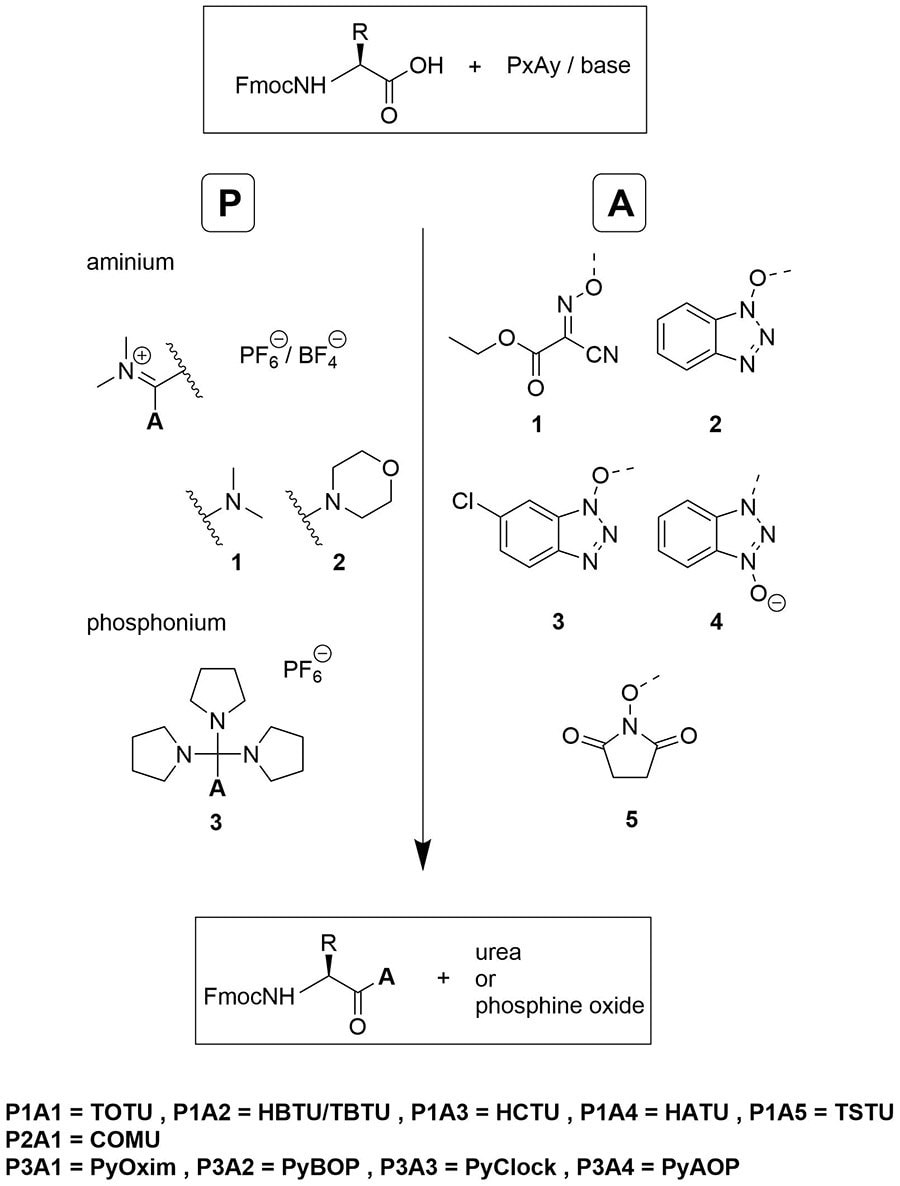
The challenge of peptide nucleic acid synthesis - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D2CS00049K

Hydrodehalogenation of Alkyl Iodides with Base-Mediated Hydrogenation and Catalytic Transfer Hydrogenation: Application to the Asymmetric Synthesis of N-Protected α-Methylamines | The Journal of Organic Chemistry

Metal and carbene organocatalytic relay activation of alkynes for stereoselective reactions | Nature Communications
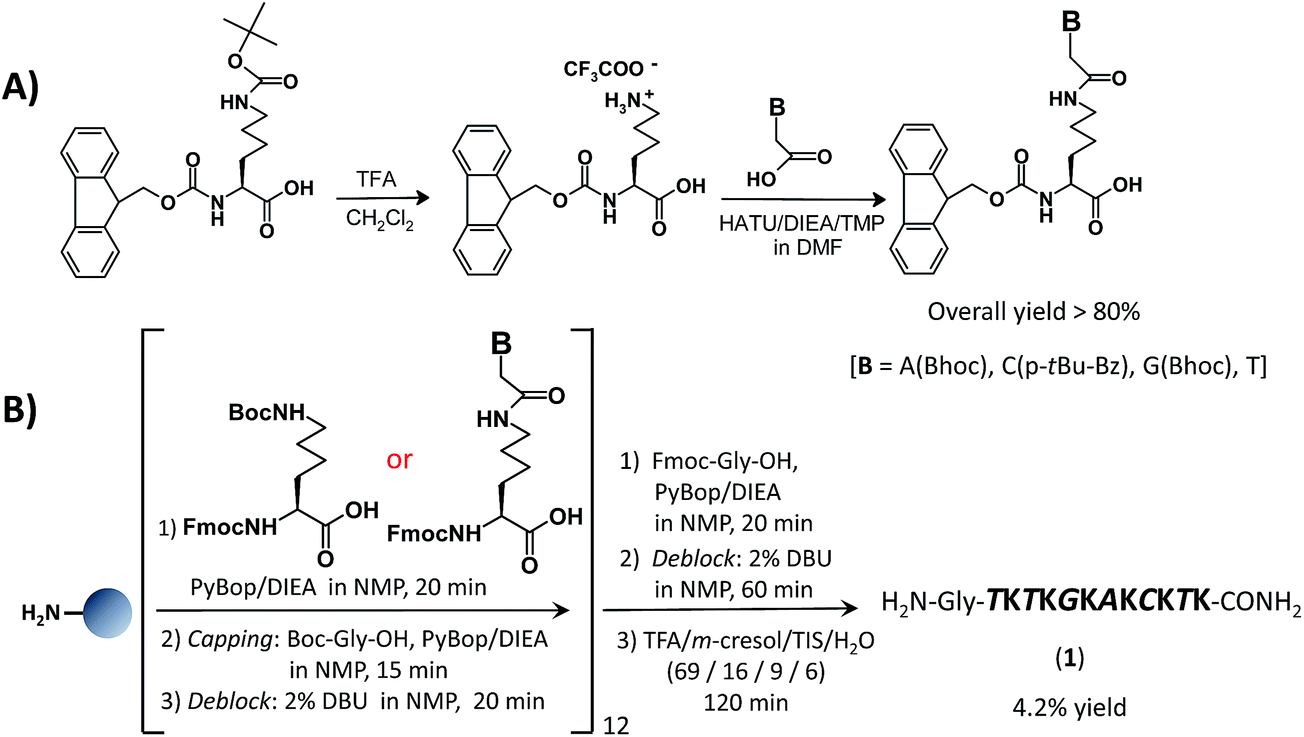
Synthetic approaches to nucleopeptides containing all four nucleobases, and nucleic acid-binding studies on a mixed-sequence nucleo-oligolysine - RSC Advances (RSC Publishing) DOI:10.1039/C6RA08765E