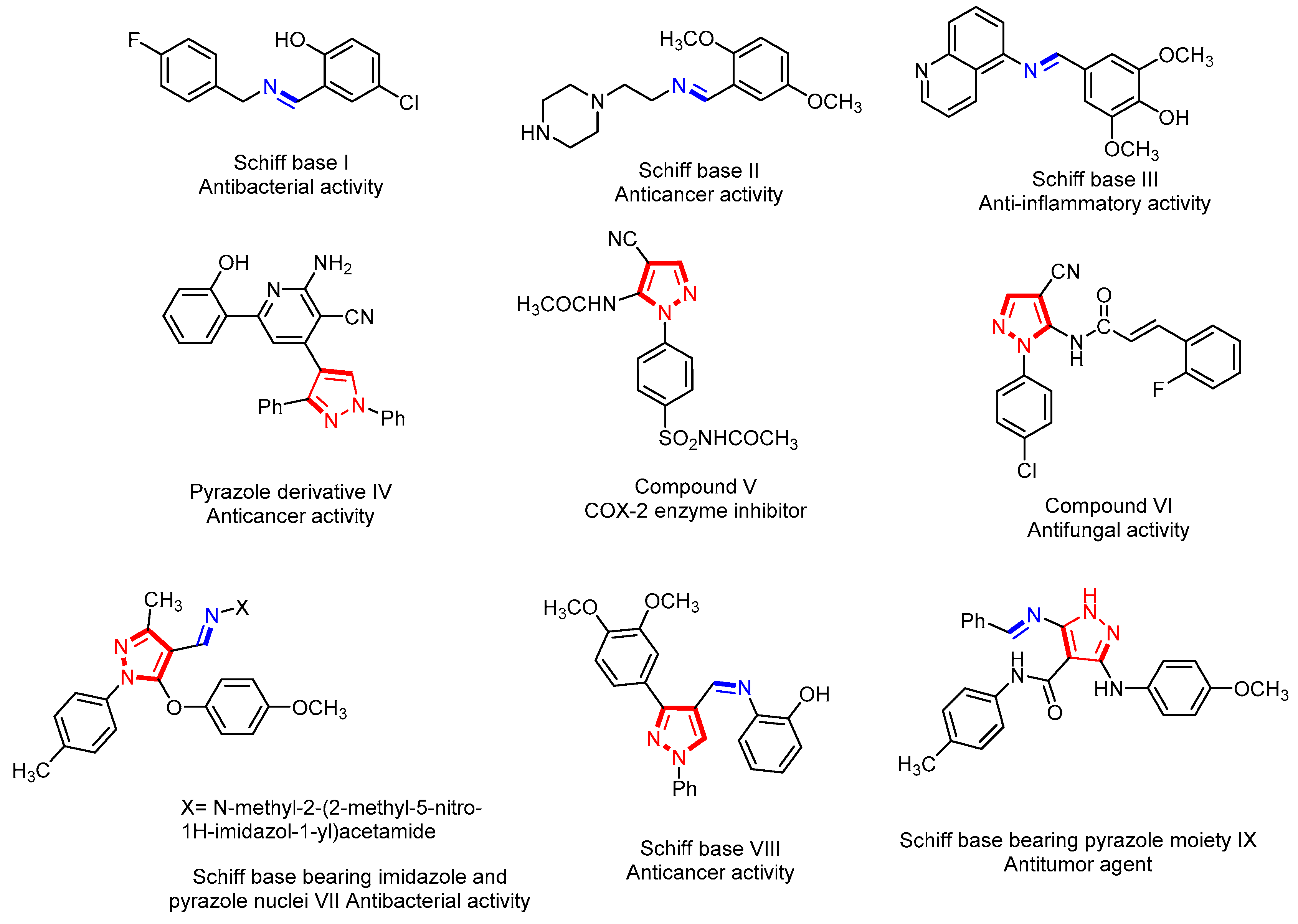
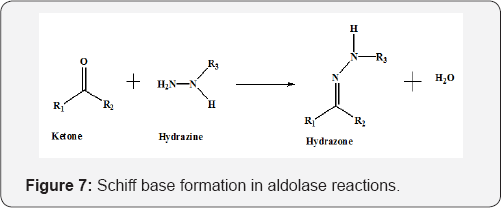
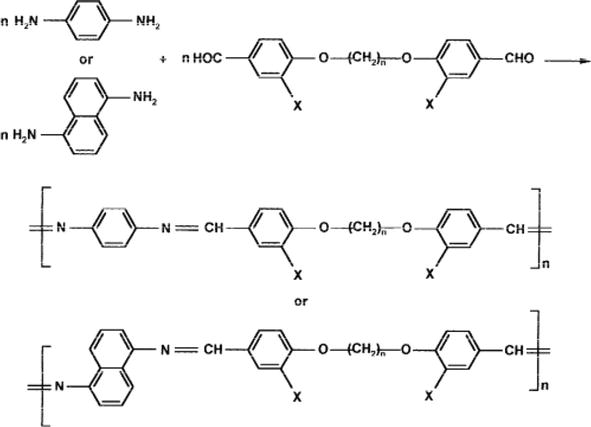
Designing metal chelates of halogenated sulfonamide Schiff bases as potent nonplatinum anticancer drugs using spectroscopic, molecular docking and biological studies | Scientific Reports

Recent Advances in the Catalytic Applications of Chiral Schiff‐Base Ligands and Metal Complexes in Asymmetric Organic Transformations - De - 2022 - ChemistrySelect - Wiley Online Library

Enhancement of Schiff base biological efficacy by metal coordination and introduction of metallic compounds as anticovid candidates: a simple overview

Amino Acid Schiff Base Bearing Benzophenone Imine As a Platform for Highly Congested Unnatural α-Amino Acid Synthesis | Journal of the American Chemical Society

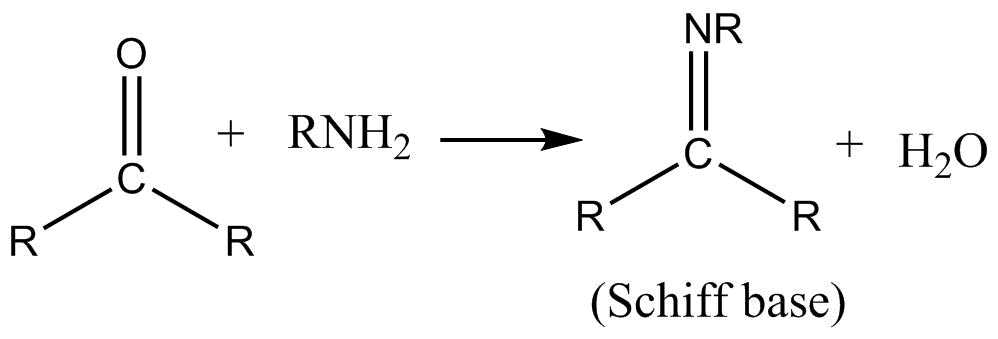
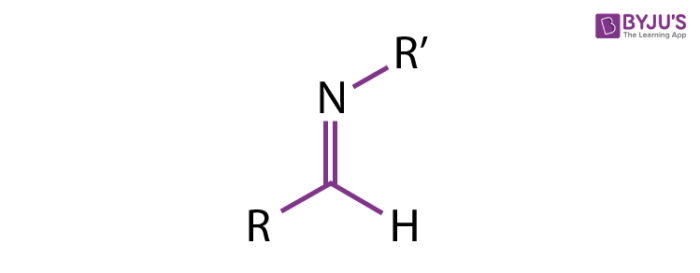
Reversible reaction of a Schiff base formed from an aldehydes or ketones. | Download Scientific Diagram
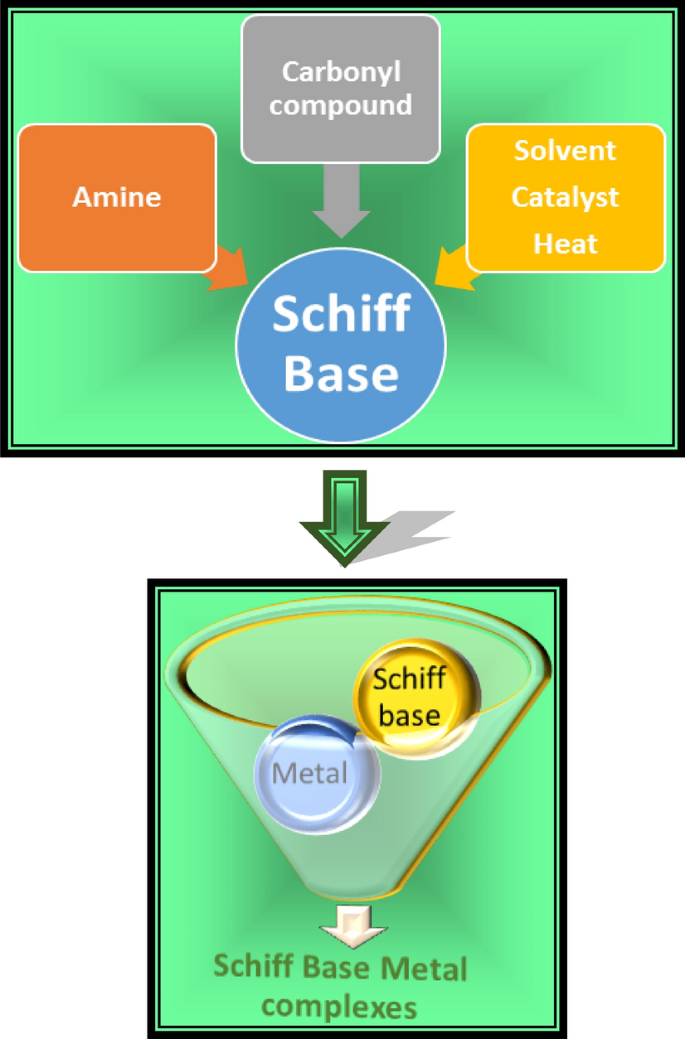
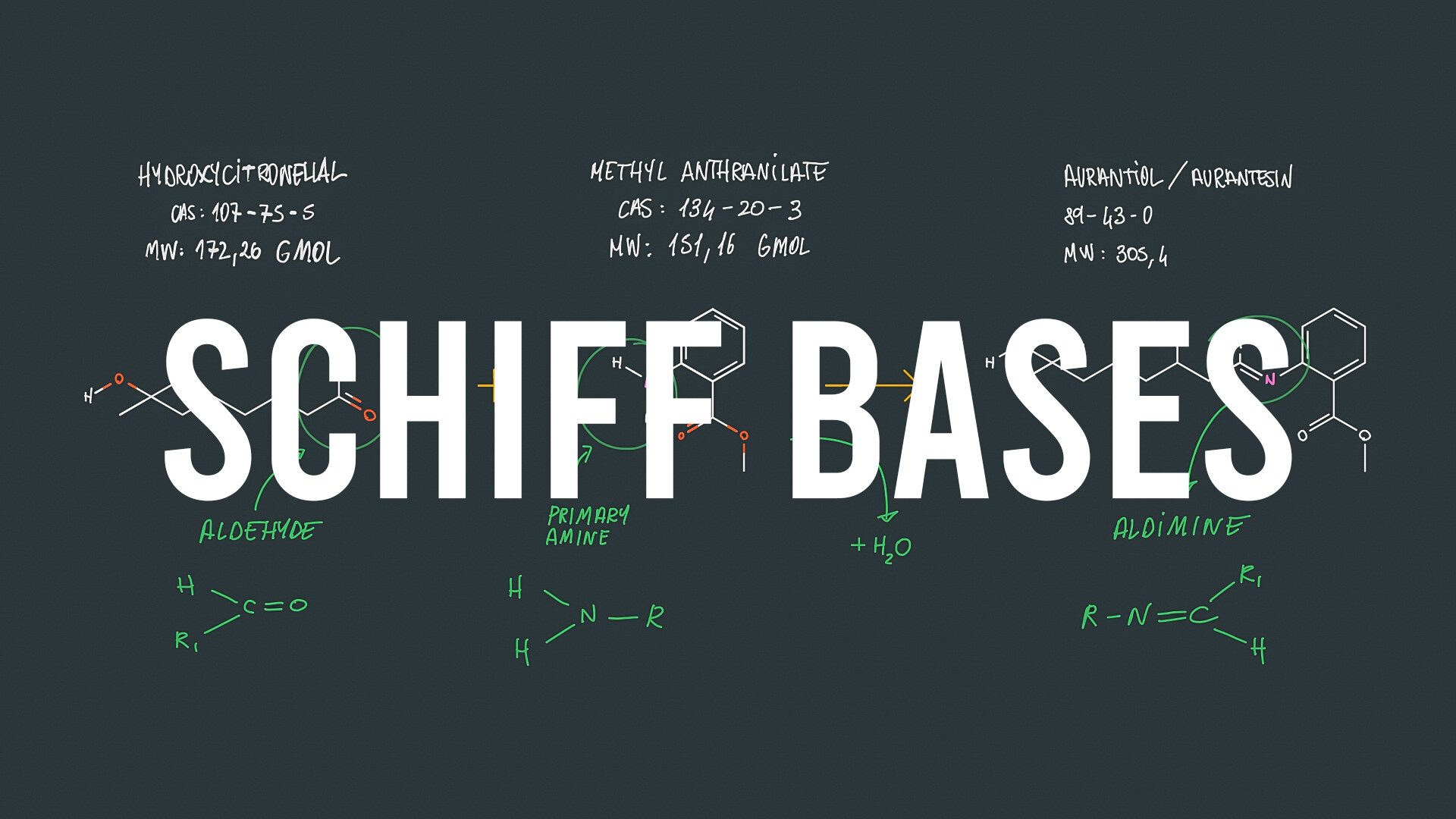
Microwave-assisted synthesis and notable applications of Schiff-base and metal complexes: a comparative study | SpringerLink
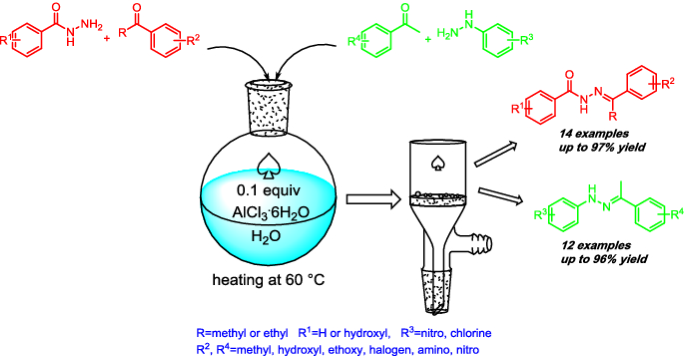
AlCl3·6H2O-catalyzed Schiff-base reaction between aryl ketones and aromatic acylhydrazines/hydrazines in water | SpringerLink

Schiff base complexes and their versatile applications as catalysts in oxidation of organic compounds: part I - Al Zoubi - 2017 - Applied Organometallic Chemistry - Wiley Online Library

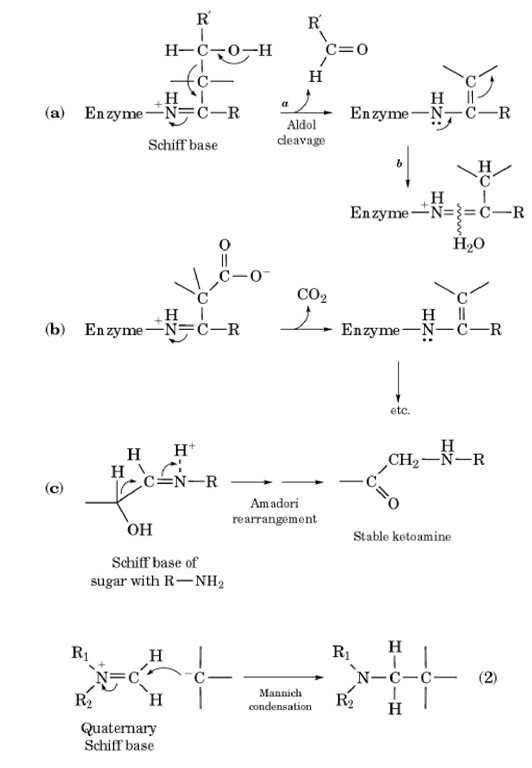
Metal complexes driven from Schiff bases and semicarbazones for biomedical and allied applications: a review | Semantic Scholar
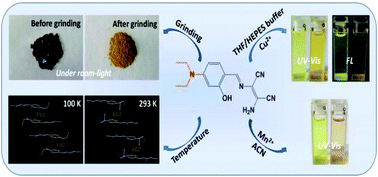
Photophysical properties of a D–π-A Schiff base and its applications in the detection of metal ions - Dalton Transactions (RSC Publishing)
![New insights into the mechanism of Schiff base synthesis from aromatic amines in the absence of acid catalyst or polar solvents [PeerJ] New insights into the mechanism of Schiff base synthesis from aromatic amines in the absence of acid catalyst or polar solvents [PeerJ]](https://dfzljdn9uc3pi.cloudfront.net/2020/ochem-4/1/fig-1-full.png)
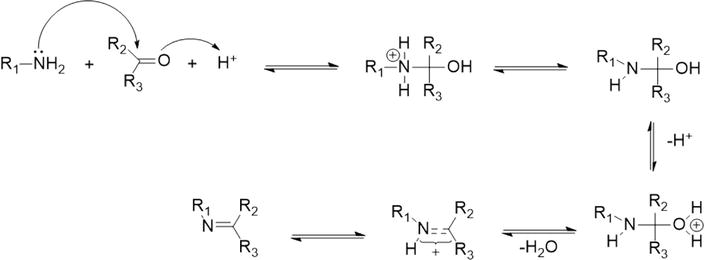
New insights into the mechanism of Schiff base synthesis from aromatic amines in the absence of acid catalyst or polar solvents [PeerJ]