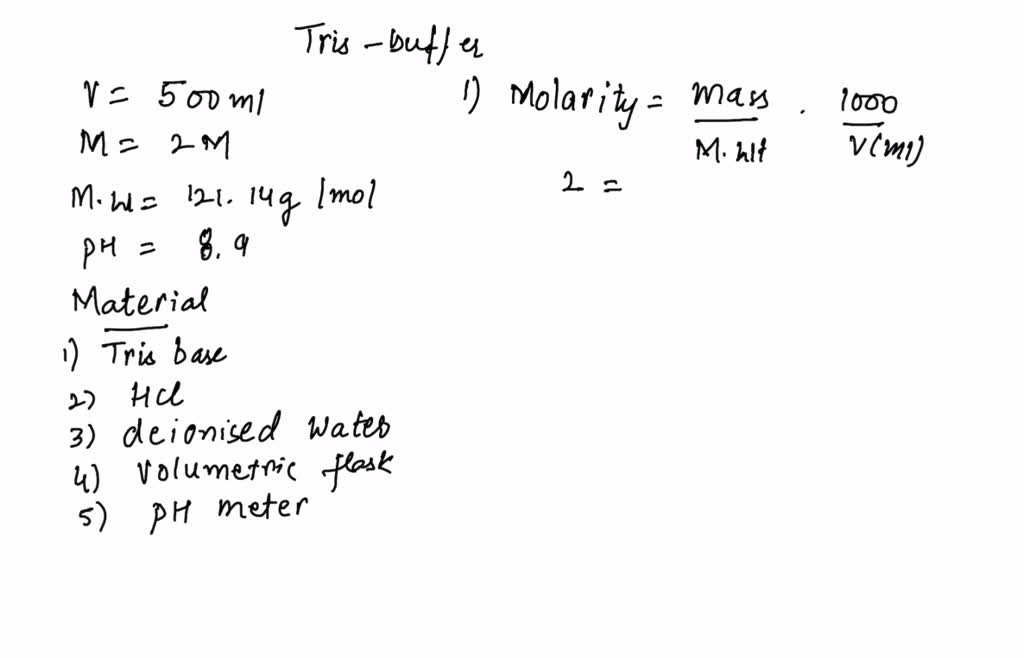
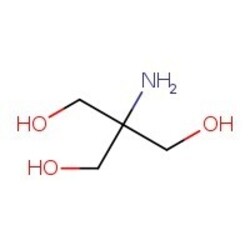
SOLVED: You have to prepare the following solution (show your work): 500 mL of 2M Tris-HCl buffer, pH 8.9. Tris base MW is 121.14 g/mol. Please describe the preparation procedure for the
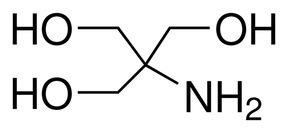
Tris Base Ultrapure (Tris(hydroxymethyl) aminomethane, 2-Amino-2-(hydroxymethyl)-1, 3-propanediol, T | CAS 77-86-1 | United States Biological | Biomol.com
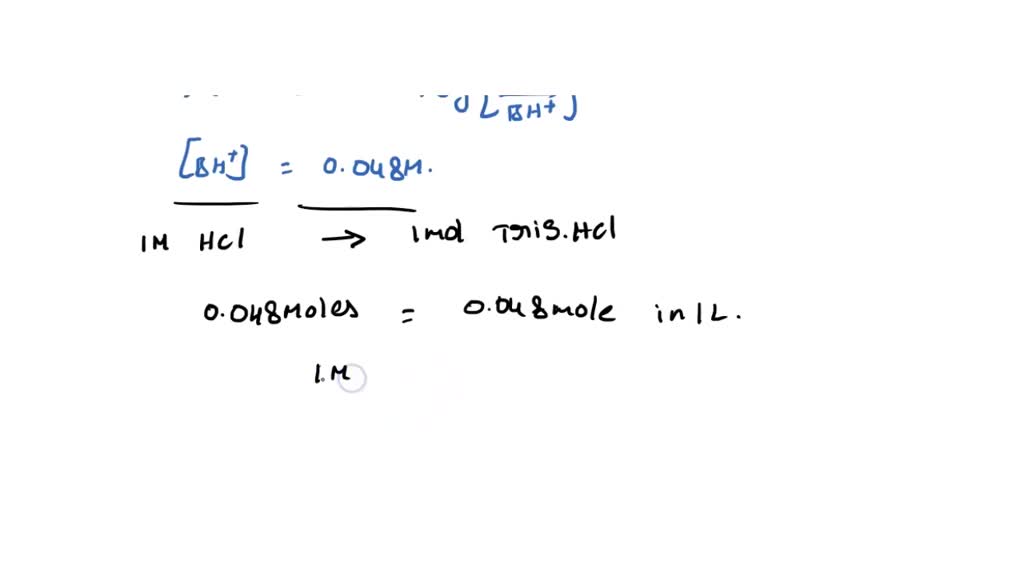

![TE Buffer [100X] TE Buffer [100X]](https://cdn.gbiosciences.com/cache/786-813%20(new)-500x500.jpg)

![G-Biosciences TE BUFFER [1X], PH 8.0, LOW EDTA (TRIS-EDTA; 10MM TRIS BASE, | Fisher Scientific G-Biosciences TE BUFFER [1X], PH 8.0, LOW EDTA (TRIS-EDTA; 10MM TRIS BASE, | Fisher Scientific](https://assets.fishersci.com/TFS-Assets/CCG/product-images/VN00037630-786-150.JPG-650.jpg)

![T60040-1000.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 1 Kilogram T60040-1000.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 1 Kilogram](https://d2gdaxkudte5p.cloudfront.net/system/images/T60040-1000.0_.jpg)











![T60040-500.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 500 Grams T60040-500.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 500 Grams](https://d2gdaxkudte5p.cloudfront.net/system/images/T60040-500.0_.jpg)