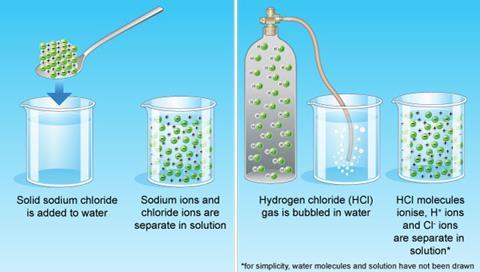
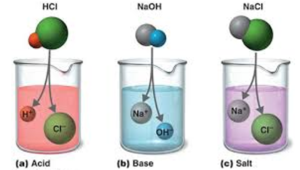
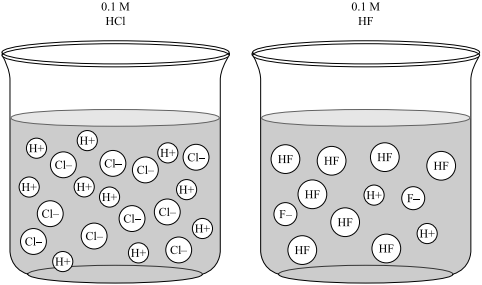
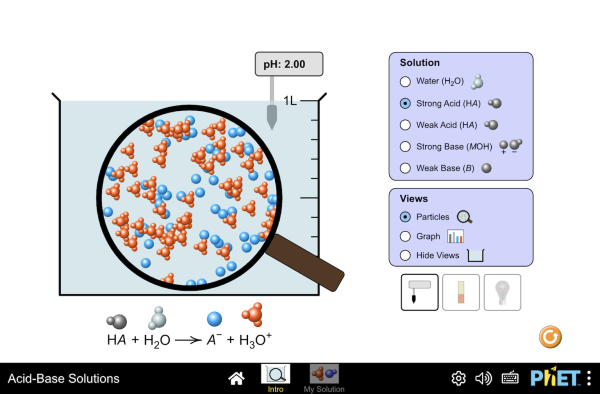
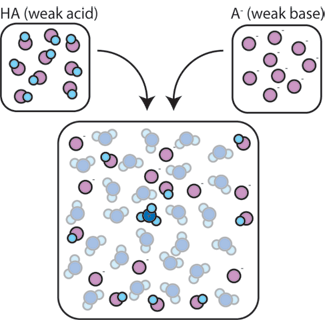
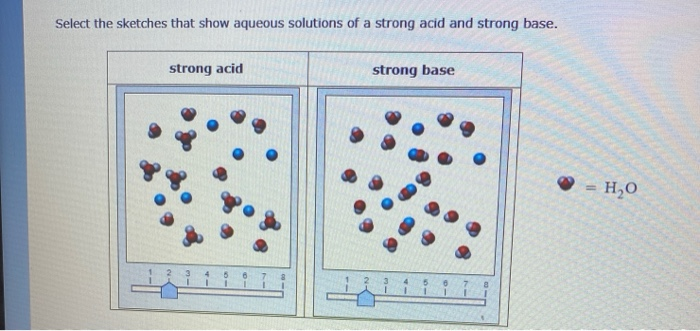
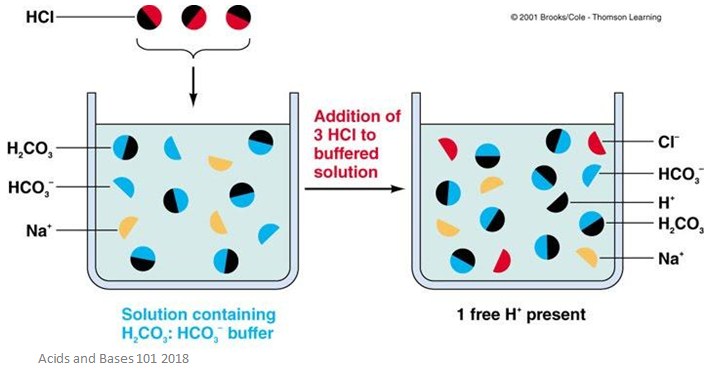
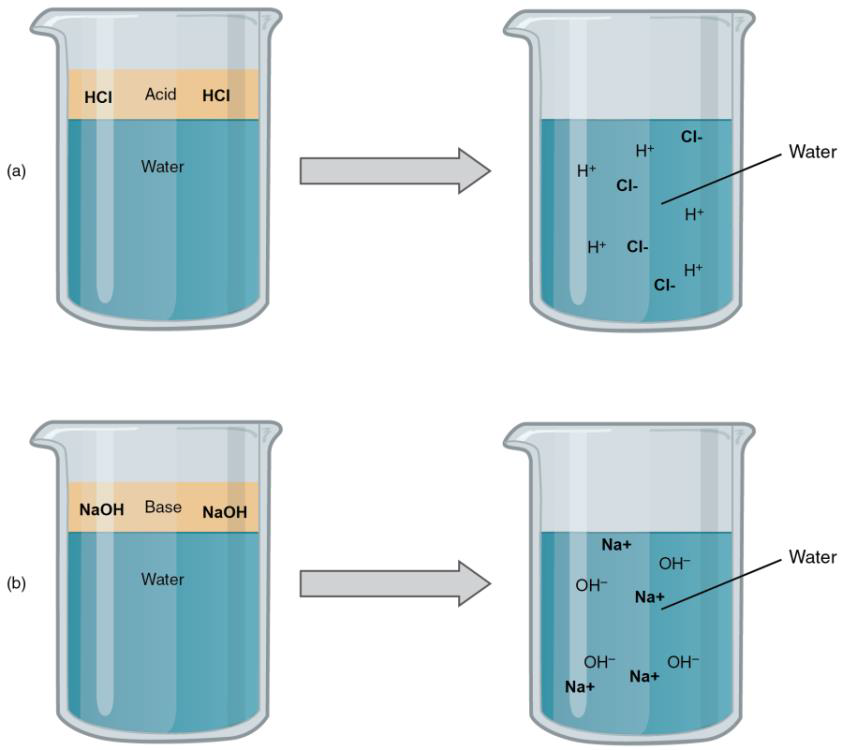
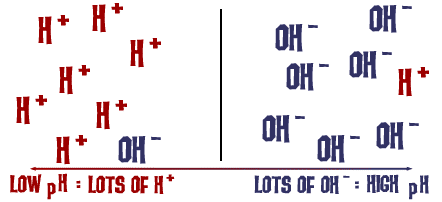
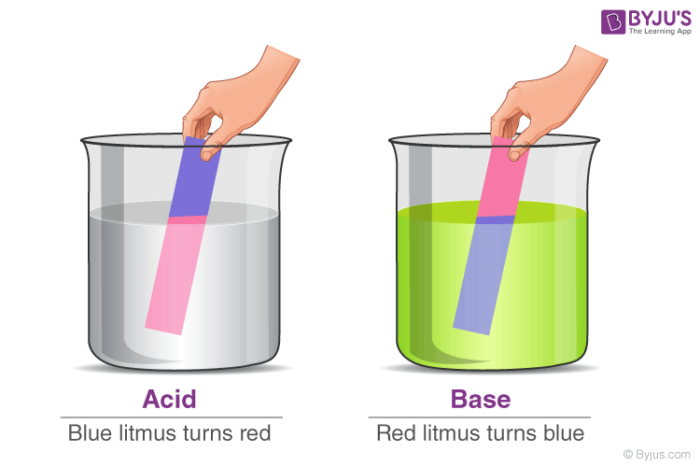
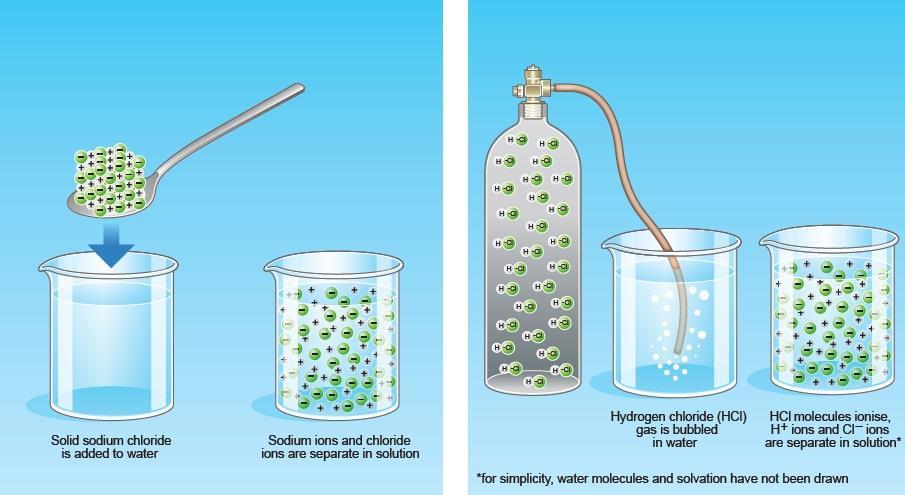
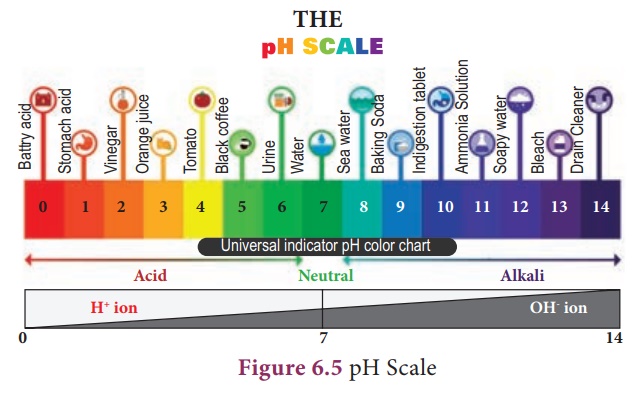
Sodium hydroxide (NaOH) is classified as a strong base. For every mole of sodium hydroxide added to a large volume of water, one mole of what ion enters the solution? | Socratic

Dimerization of HNO in Aqueous Solution: An Interplay of Solvation Effects, Fast Acid–Base Equilibria, and Intramolecular Hydrogen Bonding? | Journal of the American Chemical Society